FDA Warns Consumers Not To Purchase CVS Eyedrops Due To Risk Of Bacterial Infection
The Food and Drug Administration recently recommended CVS remove eye drops from their stores after a production area was found to be contaminated with bacteria. CVS has since issued a recall of the eye drop products. The alert came in October 2023 when investigators revealed that the eye drops could be contaminated with bacteria. In August, the FDA urged consumers to not use LightEyez MSM Eye Drops Eye Repair and Dr. Berne’s MSM Drops 5% Solution over potential fungal and bacterial contamination.
In February of 2023, the CDC reported an outbreak of Pseudomonas aeruginosa that was linked to 10 different brands of eye drops. The most common of these brands was Ezri Care Artificial Tears. The bacteria was found in 81 individuals. 4 of these individuals have died from infections, according to the CDC.
Upon investigation, the CDC determined that microbial testing by an India-based company was inadequate and that the manufacturer failed to use tamper-resistant packaging and distributed the eye drops without proper preservatives.
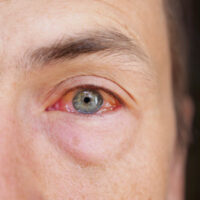
Injuries caused by the eye drops
Injuries caused by the eye drops include blurry vision, discharge from the eye, pain and discomfort, redness of the eye, a feeling like there is something in your eye, and increased sensitivity to light.
Those who experience any of those symptoms after using the eye drops may have the opportunity to file a lawsuit against the manufacturer on the grounds of product liability. These types of cases are often complex and require the aid of a skilled product liability lawyer to file. In this case, the companies that manufactured the eye drops could be on the hook for compensatory damages related to any serious injuries that were caused by the offending eyedrops.
In addition to the manufacturer being held liable for injuries caused by the eyedrops, the stores that sold them such as CVS and Target may also be held liable for these injuries. CVS has already been ordered by the FDA to pull the eye drops from their shelves as has Target.
Elements of negligence
If you sustain a bacterial infection due to contaminated eye drops, the manufacturer is liable for not preventing the eye drops from becoming contaminated. It is, therefore, presumed that their negligence is responsible for causing injury to customers. In these cases, manufacturers are strictly liable for any injuries that they cause to the public. That means you don’t necessarily need to prove negligence, but negligence allegations bolster claims against manufacturers and could lead to a finding of punitive damages which are levied against a defendant to punish them for gross negligence. In this case, manufacturers are expected to conduct routine inspections of areas where the eye drops are manufactured and to ensure that they are sanitary. In this case, the plaintiffs claim that they failed to do so.
Talk to a Florida Product Liability Lawyer Today
If you have been injured by a dangerous or defective product, call the Florida personal injury lawyers at Halpern, Santos & Pinkert today to schedule a free consultation and learn more about how we can help.
Source:
nytimes.com/article/eye-drops-recall-explained.html














